Digital therapeutics (DTx) have moved from experimental pilots to fully regulated treatment options used alongside or instead of traditional therapies. They combine medical evidence and software to help patients manage chronic conditions, improve adherence, and change behavior.
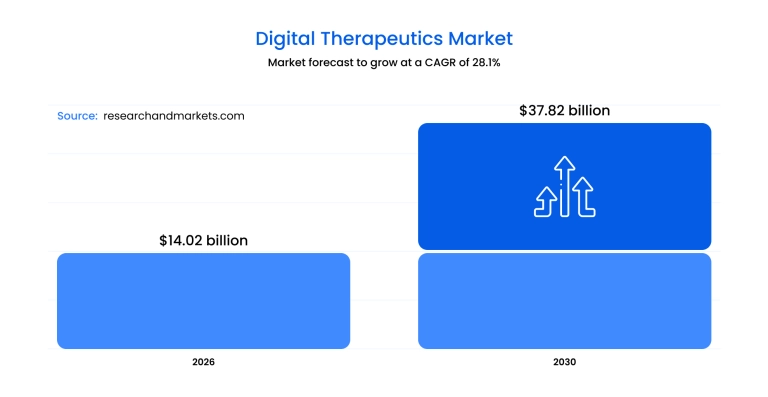
According to a 2026 industry report from Research and Markets, the sector has surged to a .02 billion valuation this year, maintaining a 28.3% CAGR. It’s a reflection of a healthcare system desperately trying to solve the problem of patient non-adherence and the rising costs of chronic care.
Building these tools is high-stakes engineering. To move from a prototype to a regulated product, leaders must overcome technical hurdles without sacrificing user experience. In 2026, success belongs to those who prioritize digital therapeutics development that proves clinical efficacy over marketing hype.
Despite this growth, the path to market remains complex. Developing successful DTx solutions requires navigating a "triad of trust": rigorous clinical validation, stringent data security, and seamless integration into the lives of patients and clinicians alike.
What are digital therapeutics (DTx)?
Digital therapeutics are evidence‑based software applications that prevent, manage, or treat medical conditions by delivering clinically validated therapeutic interventions via mobile apps, web, or connected devices. Unlike general wellness tools, these products must adhere to rigorous standards of clinical efficacy and regulatory oversight to ensure patient safety and therapeutic impact.
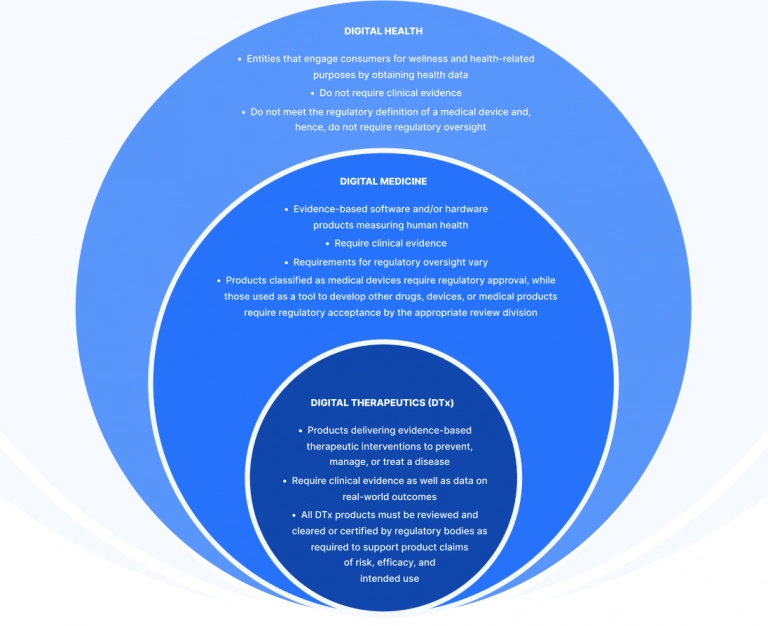
DTx full form and meaning
In the healthcare industry, DTx full form stands for digital therapeutics. In modern healthcare, the term refers to clinically validated software that delivers medical interventions, typically through mobile or web interfaces, to manage or treat specific diseases.
While the market is flooded with health apps, true digital therapeutics solutions are distinguished by their requirement to prove safety and effectiveness through peer-reviewed clinical studies.
The regulatory environment remains the ultimate gatekeeper for these technologies. It's defined by the FDA-EMA joint guiding principles on Good AI Practice, which demand that software-driven interventions maintain the same level of data integrity and patient safety as traditional pharmaceuticals.
This unified approach ensures that digital therapeutics are held to strict and harmonized standards across global markets. Bringing these products to market requires navigating a complex development lifecycle to ensure the software meets the criteria required for medical certification.
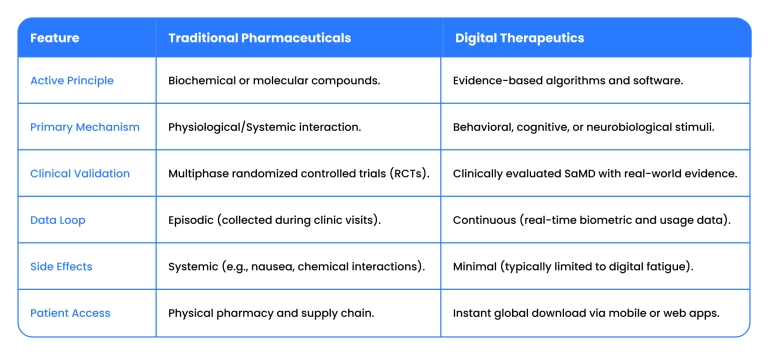
Digital therapeutics (DTx) vs traditional treatments
The relationship between digital therapeutics and pharma has evolved into a strategic alliance that addresses the clinical gaps traditional medicine cannot bridge. Unlike chemical compounds that rely on static dosing, a digital therapeutics solution uses software to drive medical outcomes through behavioral and physiological changes in real time.
In the current clinical model, the distinction between a pill and a platform is defined by three functional shifts:
- Active Intervention: Traditional drugs use biochemical pathways; DTx uses algorithmic stimuli to treat pathology directly.
- Infrastructure over innovation: The focus has moved from creating "apps" to building the secure architecture required to deliver clinical care at scale.
- Real-time titration: While pharma is "blind" between doctor visits, software provides a continuous loop of real-world evidence, allowing for immediate adjustments.
The clinical weight of these tools is now backed by significant regulatory progress. In 2024 alone, the FDA authorized an all-time high of 235 AI-enabled devices, marking a 32% increase in under a year. This surge is largely due to the digital therapeutics alliance establishing clear clinical benchmarks that move these tools into the category of "prescriptive medicine”. As experts from the Journal of Medical Internet Research highlight regarding the infrastructure challenge:
"Innovation creates tools. Infrastructure determines access. Meaningful population-level outcomes emerge only at the intersection of clinical evidence, user engagement, and scalable infrastructure”.
These categories are not rivals but a complementary forces in a modern care plan.
Here is the comparison breakdown:
Prominent digital therapeutics examples now include game-based treatments for ADHD and platforms that automate insulin titration based on biometric feedback. However, building at this level is high-stakes engineering.
To move from a prototype to a regulated product, leaders must overcome technical hurdles without sacrificing user experience. Many developers find that cloud computing in healthcare is the only way to manage the massive data loads required for these interventions without compromising security.
Digital therapeutics market outlook
In 2026, the US digital therapeutics market has transitioned from a high-growth niche into a cornerstone of the national healthcare infrastructure. Besides extreme market growth, the industry shifts toward outcome-based revenue models.
This maturation is driven by two major factors: the expansion of Medicare coverage for digital therapeutics products and the widespread clinical adoption of "software-only" interventions for chronic conditions.
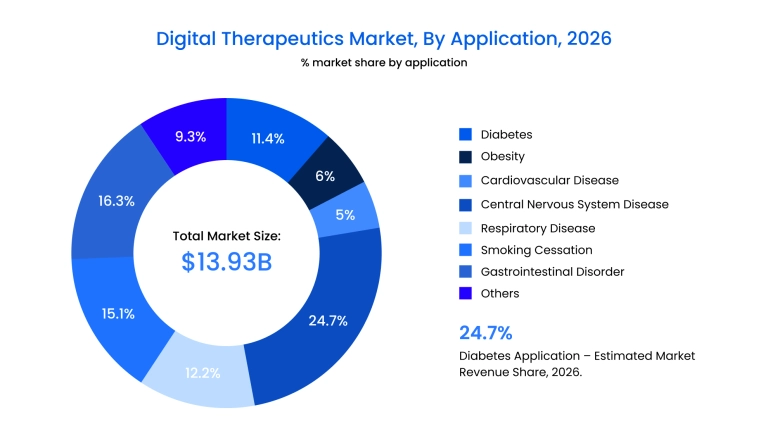
Key market drivers for 2026 include:
- The "B2B" dominance: Healthcare providers and insurers now account for nearly 48% of the market share, favoring scalable digital therapeutics solutions that integrate directly into clinical workflows.
- Therapeutic focus: Diabetes remains the largest application segment, representing 24.7% of the market, followed closely by mental health and neurological disorder management.
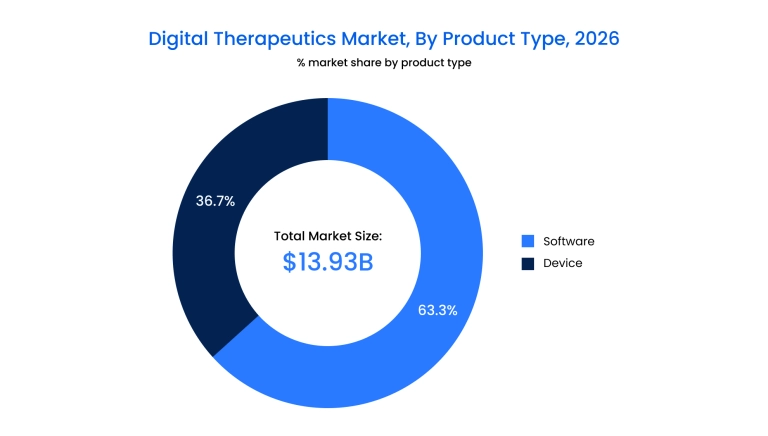
- The rise of SaMD: Software-only offerings are outpacing device-integrated tools due to their lower incremental costs and ability to be updated in real-time without physical recalls.
Digital therapeutics and public markets
The volatility that once defined digital therapeutics stock has been replaced by a focus on "operational truth". Today, investors prioritize companies that can demonstrate rigorous DTx compliance and clear reimbursement codes (such as HCPCS A9291 for cognitive therapy). Public market leaders are no longer judged on user acquisition alone. Their valuation is now anchored to clinical efficacy and their ability to withstand the hardware-layer security threats.
The benefits of digital therapeutics are now being realized at a population scale, with platforms like Canvas Dx (autism diagnosis) and Rejoyn (depression) proving that digital tools can deliver medical-grade outcomes.
For a real-world look at the engineering behind these outcomes, check out how Binariks developed a remote patient monitoring application that uses machine learning to analyze vital signs and predict health deterioration. This architecture ensures high-stakes data integrity and seamless EHR integration, providing a blueprint for scalable clinical interventions.
The public market distinguishes winners by their ability to maintain strict DTx compliance and prove long-term patient adherence.
As companies move toward public offerings, the focus has shifted to the underlying infrastructure, investors now prioritize platforms that can demonstrate high-stakes data security and seamless integration into existing EHRs. This financial stability has paved the way for more examples of digital therapeutics to become standard-of-care options for millions of patients worldwide.
Core principles of digital therapeutics
For software to be classified as a digital therapeutics solution, it must move beyond general wellness tracking and align with the rigorous "Foundational Ten" established by the industry. In 2026, these are the architectural requirements for any digital therapeutics in healthcare product.
According to the Digital Therapeutics Alliance , a true DTx product must:
- Treat, manage, or prevent: Have a primary clinical objective to deliver a software-driven intervention for a specific disease or medical condition.
- Adhere to medical standards: Be developed under strict design, manufacturing, and quality management systems (such as ISO 13485) to ensure safety.
- Generate clinical evidence: Undergo rigorous clinical trials and publish results in peer-reviewed journals to prove the intervention actually works.
- Secure regulatory clearance: Obtain certification from bodies like the FDA or EMA to legally support claims of risk, efficacy, and intended medical use.
- Apply real-world evidence: Continuously collect and analyze performance data after launch to refine the treatment and maintain DTx compliance.
- Focus on end-user engagement: Involve patients in the usability testing process to ensure high adherence, as the therapy only works if the patient uses it.
These principles ensure that digital therapeutics companies provide the same level of safety and reliability as traditional pharmaceutical interventions.
For example, Binariks developed an early heart disease detection solution that successfully bridged the gap between raw biometric data and actionable cardiovascular care.
By aligning the system with evidence-based interventions and the IMDRF's definition of medical purpose, such as the "diagnosis, prevention, monitoring, treatment or alleviation of disease", the solution moved from a basic tracking tool to a proactive, clinical-grade platform.
Real DTx use cases
The modern landscape of the digital therapeutics sector has moved far beyond simple reminders and basic logging. Today, advanced solutions operate as clinically validated interventions that use real-time data to drive behavioral change and physiological outcomes.
The following use cases demonstrate how DTx integration is currently reshaping chronic disease management and musculoskeletal care through high-tech, scalable software.
1. Cardiometabolic care: Diabetes and obesity
Diabetes and weight management have become the flagship applications for DTx, particularly with the recent surge in GLP-1 medications. Modern platforms now go beyond glucose tracking to offer "personalized digital health ecosystems".
For example, in 2025, Welldoc partnered with Eli Lilly to launch a dedicated digital support platform for patients on Mounjaro and Zepbound. These solutions use AI to provide real-time coaching on nutrition and medication adherence, helping patients manage the complex "hospital-to-home" transition.
2. AI-driven MSK: Kaia Health & Sword Health
Musculoskeletal (MSK) therapy is a primary driver of the digital therapeutics cloud infrastructure, where real-time processing is essential. In a landmark January 2026 consolidation, Sword Health acquired Kaia Health for $285 million, uniting Kaia’s markerless computer vision with Sword's clinical ecosystem.
This partnership leverages proprietary AI to analyze movement via a smartphone camera, delivering physical-therapy-grade feedback that has demonstrated a 46% reduction in pain intensity and an 81% decrease in overall healthcare costs for long-term users.
3. Chronic condition management: Binariks case study
At Binariks, we have firsthand experience in the specialized engineering required to transition wellness hypotheses into the digital therapeutics sector. In our YourCoach case study , we re-architected a health coaching platform to meet the rigorous demands of B2B DTx integration for major corporate partners like Twill.
By implementing a digital therapeutics cloud architecture with isolated data instances and an iFrame-based widget system, we enabled the platform to scale while ensuring 100% HIPAA/GDPR compliance.
Our implementation of automated reporting (generating 10+ daily metrics via SFTP) and custom scheduling engines provided the high-precision infrastructure necessary to support validated clinical results, such as a 17.9% reduction in user anxiety. This project illustrates how custom engineering can transform a medical tool into a scalable, enterprise-grade therapeutic intervention.
These use cases illustrate that the most successful DTx solutions are those that integrate seamlessly with existing medical hardware and clinical workflows. By leveraging cloud-based AI and expert engineering, these platforms provide a level of continuous, proactive care that traditional intermittent doctor visits simply cannot match.
How Binariks helps with DTx app development
To provide a concrete look at how these standards function in practice, we can look at the engineering frameworks we’ve refined through our work in the digital therapeutics sector. Our approach isn't just about writing code; it’s about building a "validated medical instrument" that survives the transition from a startup’s vision to an institutional-grade reality.
Bridging the gap from idea to FDA clearance
In our work with clients like YourCoach, we found that the most critical phase isn't the final launch, but the initial DTx app development design. We utilize a dedicated team, comprising a solution architect, business analyst, and compliance engineers, to ensure the software architecture inherently follows the HIPAA "Security Rule".
By validating ideas through the three lenses of acceptability, feasibility, and effectiveness, we help partners add complex features that require MDR or FDA approval without having to restart the development lifecycle.
Engineering for interoperability and scale
A major hurdle in diabetes digital therapeutics is making sure the software doesn't live in a silo. We focus heavily on DTx integration, building systems that "speak" to the broader healthcare ecosystem via HL7/FHIR standards. In our case studies, this involved:
- Architectural adjustment: Shifting from B2C to B2B models by implementing multi-tenant, digital therapeutics cloud environments.
- Security-first SDLC: Integrating automated vulnerability scanning and penetration testing directly into our Scrum ceremonies to ensure the product remains secure through every sprint.
- Regulatory documentation: Providing the "auditable validation" needed for faster clearance from healthcare bodies, ensuring that the technical documentation is ready for inspection at any moment.
Ultimately, our role is to act as the technical foundation that allows medical innovators to focus on science. By managing the complexities of "trust-by-design", from initial solution design to long-term maintenance, we transform medical hypotheses into scalable tools that deliver real-world clinical results.
FAQ
Share

